About Medicover Integrated Clinical Services
Medicover MICS is specialized business unit of Medicover Group offering patient-centric, global clinical trial solutions.
Medicover Integrated Clinical Services is your trusted partner for patient-centric, global clinical trial support
Backed by over 25 years of experience in clinical development, we seamlessly connect advanced central laboratory services, precision diagnostics, and the comprehensive management of our wholly owned clinical research network. Over the past decade, we have delivered more than 750 clinical trials with 10,000+ investigators in 50+ countries, covering nearly 20 therapeutic areas – from oncology, CNS, and infectious diseases to rare and orphan disorders. Our capabilities and experience drive operational excellence and ensure reliable, actionable results at every stage of the trial.
Part of Medicover Group
Medicover Integrated Clinical Services (MICS) is a strategic business unit within Medicover – a leading international healthcare and diagnostic services company founded in 1992 and listed on Nasdaq Stockholm. We strategically utilise Medicover’s assets, including laboratories equipped with state-of-the-art diagnostic technology, medical clinics and hospitals, and the expertise and commitment of a global workforce.
This synergy enables unmatched scalability, streamlined trial delivery, and access to advanced technologies that drive clinical research success.
lab tests
biosamples
investigators
clinical trials
customers
tests
Our core clinical solutions
As a one-stop shop, we provide comprehensive services within four business lines: Central Lab Services, Precision Medicine and Site Management Organization.
Central Lab
Services
Comprehensive solutions to coordinate every aspect of clinical trial laboratory services. We combine our professional experience and operational excellence to support clinical trial sponsors and CROs starting from study setup to the final submission of laboratory results.
Precision
Medicine
Complete multi-omics services to support targeted therapy and drug development programs. We provide end-to-end services including biomarker discovery, target validation, CDx assay development, commercialization, and regulatory services.
Site Management
Organisation
With access to major clinical study sites and a large network of more than 100 clinical centers, we successfully integrate digitization and innovative solutions supporting patient recruitment and the whole clinical trial journey.
Why Choose Medicover MICS?
We empower you to bring therapies to patients with greater confidence and efficiency – every step of the way.
At Medicover Integrated Clinical Services, we deliver an integrated clinical trial ecosystem designed to accelerate and de-risk your development programs. Our global central laboratory services, advanced diagnostics, and precision medicine solutions are combined with the management of our wholly owned clinical research network, supporting investigator and patient recruitment. We also provide end-to-end companion diagnostics development, commercialisation, and implementation.
Make your clinical trials easier
Medicover MICS History
Global Central Laboratory Network
Medicover laboratories and strategic partner facilities worldwide.
Move the map
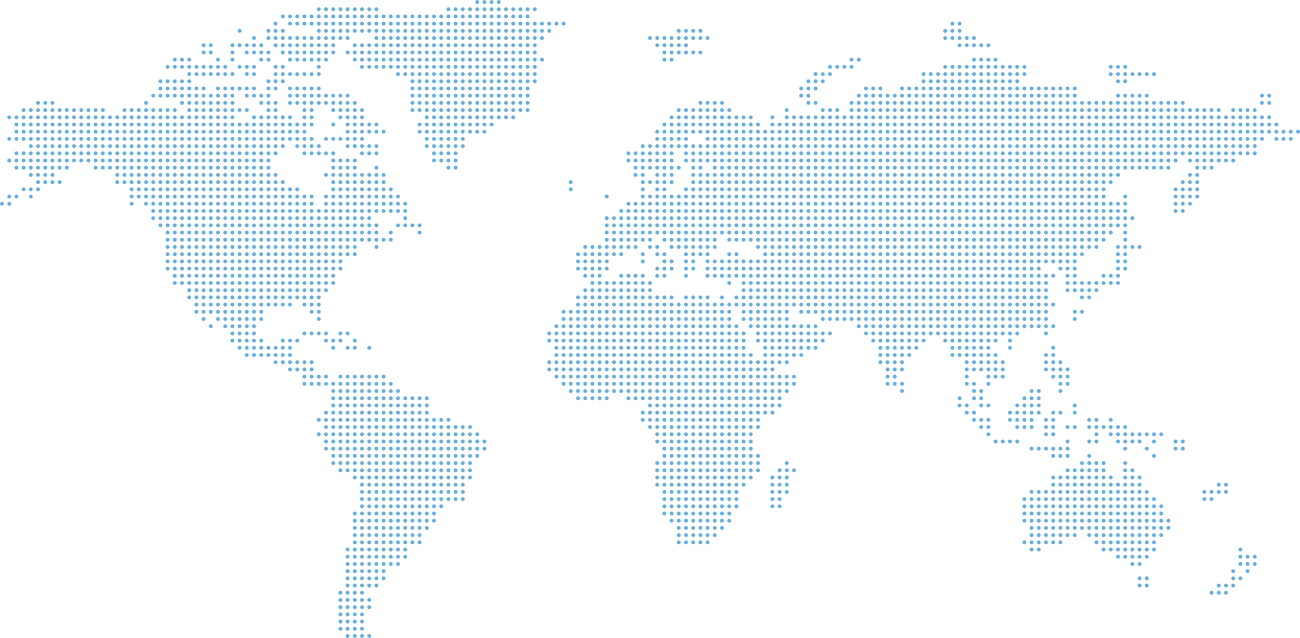
Europe
Germany — Berlin, Munich
Poland — Gdańsk, Łódź, Warsaw
Romania — Timisoara, Chiajna
Ukraine — Kiev
Sofia — Bulgaria,
Georgia — Tibilisi
Moldova —Chisinau
Turkey — Istanbul
Serbia — Belgrade
Bosna Herzegovina — Sarajevo
Netherlands — Amsterdam
UK — Handforth
Argentina
Buenos Aires
India
Mumbai
Australia
Sydney
South Korea
Yongin-si
China
Shanghai
USA
Jonhson City
Medicover MICS Management

Apostolos Siscoglou
Head of Medicover MICS
Apostolos Siscoglou brings over 15 years of international experience having held senior roles in multinational healthcare and life sciences companies, with expertise in diagnostics, genomics, and clinical trial services. He has successfully driven growth and transformation across EMEA, LATAM and global markets, working with leading pharmaceutical and biotech partners to advance clinical development and patient access to innovative diagnostics.

Łukasz Hubisz
Director, Business Unit: Central Lab Services
Łukasz Hubisz specializes in scaling businesses by aligning finance, operations, and technology. With broad experience in FP&A/controlling, IT, and program delivery, he enables fast, data-driven decision-making. He has a proven track record of building high-performing teams, leading change, and delivering results on time and within budget.

Ernest Karaś
Financial Director MICS
Ernest Karaś is an experienced finance leader with over 10 years in corporate financial management and auditing. He specializes in budgeting, FP&A, reporting and supporting strategic decision-making across complex organizations. With a proven track record in leading controlling teams and driving financial transformation, Ernest focuses on delivering operational efficiency and business growth through data-driven insights.

Marta Jeka
Global SMO Head, Business Unit: Site Management Organisation
Marta Jeka is a strategically oriented leader with experience in clinical trial operations since 2011. She has proven expertise in project management, regulatory compliance (ICH-GCP, FDA, EMA), and process optimization. Currently leading the operational management of a growing network of clinical trial centers in Poland and Germany, she is responsible for financial performance, business development, and team leadership.

Hari Radhakrishnan, PhD
Director, Business Unit: Precision Medicine
Hari Radhakrishnan brings over 10 years of experience in genetic testing and precision medicine. He specializes in companion diagnostics, supporting clients from biomarker discovery and assay development through clinical validation and commercialization. With a strong background in translational research and regulatory strategy, Hari focuses on enabling personalized therapies by integrating advanced molecular technologies into clinical practice.

Irina Ghiță, LL.M, MSc., MRQA
Director of Compliance and Quality Assurance (CQA)
Irina Ghita is a Compliance and Quality Assurance leader with extensive experience in GCP/GCLP auditing, IVDR compliance, and risk management across clinical and laboratory environments. Passionate about strengthening quality systems and ensuring excellence in regulatory alignment and operational integrity, driven by building and empowering internal teams to deliver sustainable compliance and operational success.
Our clients’ success stories
We strictly adhere to our privacy policy and, in the best interest of our clients, we cannot share all customer data. However, if you would like more details about a reference or case study, please feel free to contact us. We would be happy to connect you.
Compliance and Quality Assurance

The Compliance and Quality Assurance (CQA) Department of Medicover Integrated Clinical Services is independent from operational departments and reports directly to our Managing Director. The CQA Department is responsible to ensure that the Organisation adheres to relevant laws, regulations, and internal policies and helps safeguard the organization’s reputation and to promote a culture of integrity and ethical conduct within the Company.
The CQA personnel keep continuous contact with each MICS unit and department to ensure the same level of compliance, quality and consistency across all our units. We continually monitor our service and operations to assure that the highest level of compliance and quality is maintained.
We have implemented an Integrated Quality Management System based on the well-recognized international quality standards and regulatory guidelines such as Good Clinical Practices (ICH GCP), Good Clinical Laboratory Practices (GCLP), ISO 15189:2022, ISO 17025:2017. Additionally, our Clinical Trials Laboratory Management System (CTLMS) is compliant with Title 21 Part 11 of the Code of Federal Regulations (21CFR Part11), which applies to electronic records and electronic signatures.
We also invest significant time and resources in our employee training and operations improvement programs.
Our integrated quality management system includes:
About Medicover
Medicover Integrated Clinical Services is a unit within Medicover , dedicated exclusively to supporting drug development programs, especially clinical research. Medicover is a leading international healthcare and diagnostic services company, dedicated to delivering high-quality, patient-centric care across Europe and beyond. With an extensive network of hospitals, clinics, laboratories, and diagnostic centres, Medicover empowers Medicover Integrated Clinical Services to tap into robust clinical infrastructure and advanced diagnostic capabilities. This synergy ensures unmatched scalability, streamlined trial delivery, and access to cutting-edge technologies that drive clinical research success.
To learn more about Medicover Group, please visit their website:
www.medicover.com.
Medicover mentions in the media
Frontage Laboratories and Medicover Integrated Clinical Services Announce Strategic Collaboration to Expand Global Genomic Testing Services
- Frontage Laboratories, Inc.: Frontage Laboratories and Medicover Integrated Clinical Services Announce Strategic Collaboration to Expand Global Genomic Testing Services
- Frontage teams with Medicover for genomics services
- Frontage Laboratories and Medicover Integrated Clinical Services Announce Strategic Collaboration to Expand Global Genomic Testing Services – PharmiWeb.com
- Frontage Laboratories and Medicover Integrated Clinical Services Announce Strategic Collaboration to Expand Global Genomic Testing Services – BioSpace
Medicover MICS acquires one of Poland’s largest medical services and clinical research providers
About Medcover MICS in Googl Clinical Practice Network
Medicover Diagnostics opens new laboratory in Sarajevo
Medicover acquires IFLb Group in Berlin
Cooperation of MICS and ARUP Laboratories in companion diagnostic
Awards & Memberships of Medicover units
Work with us
We are dynamically growing, expanding our worldwide capabilisties. We are committed to fostering a globally diverse workforce and a companywide culture that encourages excellence, leadership, innovation and a balance between our personal and professional lives.
Join our team and actively contribute to the advancement of innovative medical research. We will work together to challenge the status quo and make health care better for more people, in more ways than ever. You will be part of culture that prizes innovation that impacts quality, accessibility and affordability of care across the world.
Current vacancies
HR Coordinator
Localisation: Gdańsk, Poland
Corporate news
Explore the zone for the latest updates from Medicover Integrated Clinical Services (MICS). Stay informed about our collaborations, acquisitions, and upcoming events organized by us.
Frequently Asked Questions
Contact
If you’re planning a clinical trial, have questions about our lab capabilities, or are looking for a reliable partner – feel free to contact us via the options below
We respond within 24 hours.