|
Getting your Trinity Audio player ready...
|
From vision to practice
In a previous article, we explored artificial intelligence in clinical trials from a high-level perspective, outlining the five areas of clinical development ready for AI transformation. We discussed where AI already delivers measurable value, including implementation examples, cancer detection use cases, and applications in lung and prostate cancer treatment. We also examined where AI brings the greatest impact across the clinical trial lifecycle.
You can read the full article here: AI in clinical trials
This article takes the next step. Instead of focusing on why AI matters in clinical research, we look at AI tools for clinical trials that are already being evaluated or applied in practice, as well as AI-enabled methodologies that are reshaping how trials are validated and designed.
How AI addresses key challenges in clinical trials
Clinical trials continue to face persistent challenges: slow recruitment, lengthy screening processes, rising costs, and difficulties in generating timely, high-quality data. Traditional optimization efforts often address these issues in isolation, shifting bottlenecks rather than resolving them.
AI-driven approaches act across multiple stages of the trial lifecycle simultaneously.
In patient recruitment, AI-supported matching and eligibility assessment tools analyse large volumes of structured and unstructured clinical data. Reported results show that such approaches can:
- increase enrolment rates by over 80%,
- reduce screening time by approximately 40%,
- expand the eligible patient pool by more than 100%.
During trial execution, AI-enabled digital workflows reduce operational friction and administrative burden, supporting better protocol adherence and patient retention.
AI also impacts trial outcomes and evidence generation, enabling approaches such as real-world data emulation, synthetic control arms, and more pragmatic trial designs.
Taken together, these examples show that AI does not optimise isolated tasks. By improving recruitment, execution, and outcome generation in parallel, it addresses structural inefficiencies that have long limited clinical trial performance.

This shift is no longer theoretical. A growing number of AI tools for clinical trials and AI-enabled methodologies have moved into real-world evaluation or operational use.
AI tools for clinical trials already used in practice
PyTrial – predicting trial success earlier
PyTrial is an open-source Python framework designed to predict clinical trial outcomes at both the patient and trial level.
Its capabilities include:
- patient-level predictions (response, completion, toxicity),
- trial-level outcome forecasting,
- success prediction using the SPOT algorithm (Statistical Prediction of Trial Outcomes).
Reported performance indicators show:
- >20% improvement in Phase I prediction accuracy,
- ~9% improvement in Phase II,
- ~5% improvement in Phase III.
The value of PyTrial lies in enabling earlier go/no-go decisions and reducing late-stage attrition.
TrialMatchAI – end-to-end patient matching
TrialMatchAI focuses on automating patient-trial matching by processing both:
- structured data (e.g. laboratory values, demographics), and
- unstructured data (e.g. clinical notes, pathology reports).
Key capabilities include:
- criterion-level eligibility checks with explainability,
- traceable, clinician-friendly recommendations,
- modular, secure deployment aligned with GDPR and HIPAA requirements.
In practice, TrialMatchAI enables pre-screening of around 1,000 patients in hours rather than days or weeks, significantly reducing manual workload at study sites.
Trial Pathfinder – broadening eligibility safely
Restrictive eligibility criteria remain a major barrier to recruitment, often driven by precedent rather than evidence.
Trial Pathfinder addresses this by:
- analysing historical clinical trial data,
- modelling safety outcomes linked to specific eligibility criteria,
- identifying criteria that can be safely relaxed,
- simulating the impact on patient eligibility.
Published results demonstrate an average 107% increase in the eligible patient pool without compromising patient safety.
AI-enabled trial methodologies: from validation to design
AI is not only introducing new tools, but also enabling new methodological approaches that influence how trials are validated and designed.
Silent trials – validating AI before it influences care
Silent trials refer to a validation approach in which AI models are evaluated under real-world clinical conditions without influencing operational or clinical decision-making.
AI systems generate predictions in parallel to standard workflows. These predictions are later compared with actual clinical decisions and outcomes, allowing teams to assess robustness, generalisability, bias, and workflow compatibility before activating AI-driven interventions.
Silent trials are increasingly used as an intermediate step between retrospective model development and prospective deployment, particularly where patient safety, regulatory expectations, and operational complexity intersect.
Synthetic control arms – an AI-enabled trial design approach
Synthetic control arms are not standalone AI tools, but AI-enabled trial design methodologies.
Instead of enrolling large randomized control groups, synthetic control arms are constructed using historical clinical trial data and real-world datasets. AI-assisted feature selection and statistical techniques such as propensity score matching are used to create comparator cohorts closely aligned with the treatment population.
When applied appropriately, this approach can:
- reduce the number of patients required in control arms,
- shorten enrolment timelines by 6–12 months,
- lower overall trial costs by 30–50%,
- improve ethical balance by assigning more patients to active treatment.
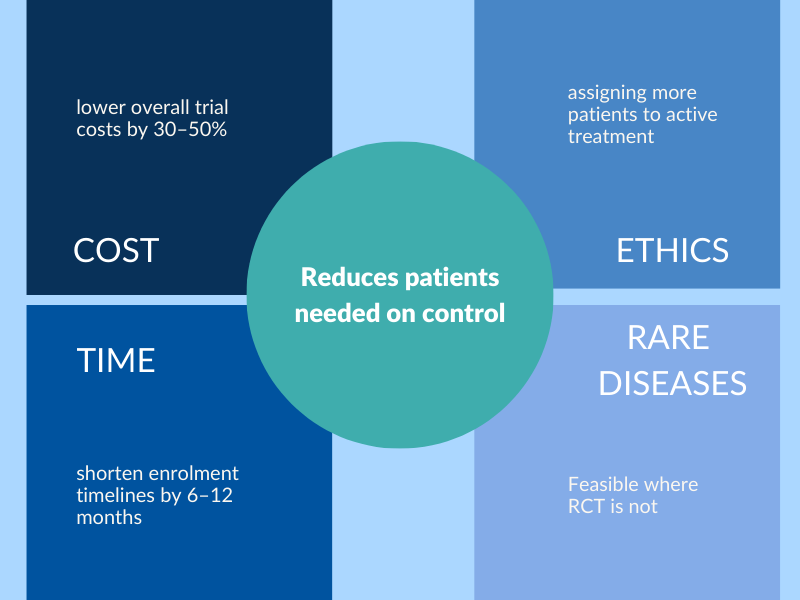
Regulatory authorities have accepted synthetic control arms in specific contexts, including rare diseases, oncology, and situations where traditional randomization is ethically or operationally challenging.
Key takeaways
AI tools for clinical trials are already delivering measurable impact. The data shows where value is created today.
- Baseline reality
Late-phase trials exceed USD 70M, last 4+ years, and ~80% miss enrolment targets. - Recruitment impact
AI-supported matching delivers:- >80% higher enrolment,
- ~40% shorter screening,
- >100% larger eligible patient pools.
- Earlier go / no-go decisions
Trial outcome prediction improves success forecasting by:- >20% (Phase I),
- ~9% (Phase II),
- ~5% (Phase III).
- Smarter trial design
AI-enabled approaches such as synthetic control arms reduce:- enrolment timelines by 6–12 months,
- total trial costs by 30–50%.
- Execution matters
AI value scales only with digital trial execution capabilities. - Data is the enabler
Advanced AI models rely on continuous digital data streams such as ePROs (electronic patient-reported outcomes) and wearable sensors.
References
- Wang Z., PyTrial: Machine Learning Software and Benchmark for Clinical Trial Applications, arXiv, 2023, [access date: 29.12.2025]
- Abdallah M. et al., TrialMatchAI: Explainable AI for Clinical Trial Matching, arXiv, 2025, [access date: 29.12.2025]
- Liu R. et al., Trial Pathfinder: Evaluating eligibility criteria of oncology trials using real-world data and AI, Nature, 2021, [access date: 29.12.2025]
- Kwong J. C. C. et al., The silent trial – the bridge between bench-to-bedside clinical AI applications, 2022, [access date: 29.12.2025]
- Basch E. et al., Effect of Electronic Symptom Monitoring on Patient-Reported Outcomes Among Patients With Metastatic Cancer: A Randomized Clinical Trial, JCO; JAMA, [access date: 29.12.2025]
FAQs – AI tools for clinical trials
1. What are AI tools for clinical trials?
AI tools for clinical trials are software solutions and analytical frameworks that use machine learning and advanced analytics to support recruitment, eligibility assessment, trial design, and decision-making.
2. Are AI tools for clinical trials already used in real studies?
Yes. Several AI tools for clinical trials are already being evaluated or applied in live research environments, particularly in oncology and rare disease studies.
3. How do AI tools improve patient recruitment?
They analyse structured and unstructured clinical data to identify eligible patients faster, increasing enrolment rates and reducing screening time.
4. Are synthetic control arms AI tools?
No. Synthetic control arms are AI-enabled trial design methodologies rather than standalone tools.
5. What are silent trials?
Silent trials are a validation approach where AI models run in parallel to standard workflows without influencing decisions, enabling safe real-world evaluation.
6. What is required to implement AI tools for clinical trials successfully?
Validated models, digital trial execution capabilities, high-quality data, and alignment with regulatory and operational requirements.






























