Since there are so many processes and mechanisms that go into operating a clinical trial, CROs or sponsors will always outsource some parts of running and managing the study through central lab. While you can expect that the prospective CRO will subcontract some of the work, it is important to find out which exact parts of the clinical study will be outsourced and it is good to know your vendors and its central lab potential. Understanding the specific capabilities and expertise of the central lab can help ensure that the quality and integrity of the trial are maintained throughout the study.
The importance of Central Lab expertise in international clinical trials
If you are planning on operating an international clinical trial, it is recommended to work with a vendor that has extensive experience in this area. While many CROs will offer near-global coverage, the level of experience with specific geographic locations can significantly vary from one vendor to another. The complexity and diversity inherent in international clinical trials necessitate the involvement of vendors with the expertise and experience to navigate the unique challenges associated with conducting trials on a global scale.

It is important to work with a CRO and vendors who has experience running clinical trials in the specific countries and regions you are planning to conduct your research in. Being compliant with the local rules and regulations for clinical testing is a very complex process that requires existing understanding and familiarity in order to ensure logistical smoothness and to mitigate legal risks. In operating a clinical trial, there are a multitude of clinical services and processes, which can greatly vary across the many regions in which you can conduct clinical testing.
A CRO that is lacking experience in operating international trials or operating in particular regions where you plan on conducting research may not be able to meet your desired quality and agility expectations, and therefore may not be the right fit for your international clinical trial. Having a vendor with experience in international clinical trials is crucial for three main reasons:
- Regulatory Compliance: Different countries have varying regulatory requirements for clinical trials. Medicover Integrated Clinical Services is a vendor with international experience that understands these regulations, ensuring that the trial processes comply with the specific requirements of each country involved. This expertise helps prevent potential delays and legal issues, ensuring a smoother trial process.
- Logistical Challenges: Managing logistics for a clinical trial across borders can be complex. An experienced vendor can navigate the challenges related to shipping, storage, and transportation of investigational products, biological samples, and other materials. The Medicover Integrated Clinical Services Logistic Team is familiar with customs regulations, and import/export requirements, and can help streamline these, sometimes challenging, processes. Their proficiency in handling logistical issues ensures timely delivery and integrity of trial materials.
- Multilingual Capabilities: Communication is key in clinical trials, and having a vendor with multilingual capabilities is advantageous. Effective communication with local investigators, regulatory bodies, and participants in their native languages can significantly enhance the accuracy and efficiency of the trial.
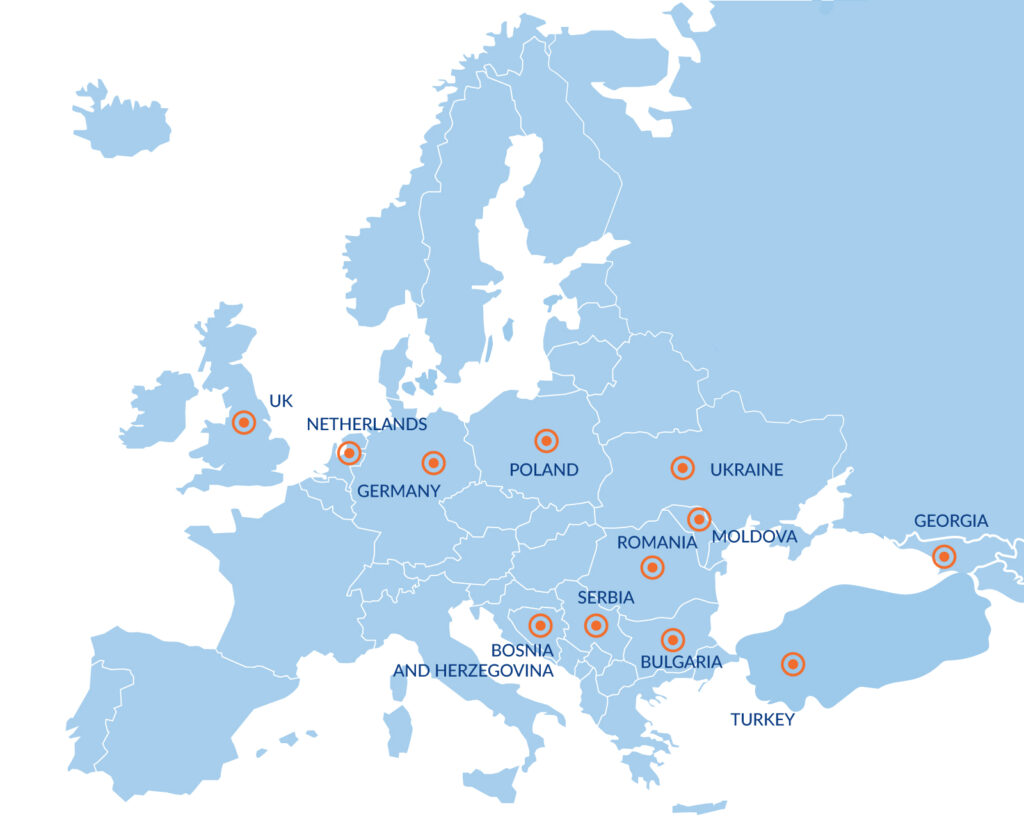
With Medicover’s strong presence in 12 countries across Central and Eastern Europe (CEE), we are uniquely positioned to support clinical trial needs. Our integrated approach covers comprehensive laboratory services and patient recruitment through our Site Management Organization. We offer an expansive portfolio of over 8000 laboratory parameters, including PK, PD, genomics, genetics, biomarkers, and PBMC isolation, catering to the diverse needs of your studies.
FAQ
1. Why should I consider a Central Lab with international clinical trial experience?
Vendors experienced in international studies understand varying regulatory environments, logistical complexities, and communication needs across multiple countries. This minimizes delays, improves operational efficiency, and helps maintain data integrity throughout the trial.
2. How do regulatory differences across countries affect Central Lab selection?
Each country has its own clinical trial regulations, documentation requirements, and approval timelines. A vendor familiar with local frameworks ensures compliance, mitigates legal risks, and prevents administrative setbacks.
3. What logistical challenges can impact global clinical trials in Central & Eastern Europe?
Cross-border transport, customs clearance, cold-chain requirements, and timely sample delivery are common challenges. Central Labs with established logistics networks can ensure sample integrity and on-time processing.
4. Why are multilingual capabilities important for a Central Lab partner?
Accurate communication with investigators, ethics committees, courier partners, and study sites in native languages improves efficiency, reduces misunderstandings, and supports smoother regulatory interactions.
5. What laboratory capabilities should I look for when selecting a Central Lab?
Sponsors should verify availability of a broad assay menu (e.g., PK, PD, biomarkers, genomics), high-capacity testing infrastructure, and specialty services such as PBMC isolation. A wide portfolio reduces reliance on additional vendors.
6. How does regional presence benefit clinical trial operations in Central & Eastern Europe?
Vendors with their own laboratories and collection sites across multiple CEE countries can streamline patient recruitment, shorten transport pathways, and enhance sample turnaround times – ultimately improving trial timelines.
Author of this article
Izabela Juszko, Business Development Manager at Medicover Integrated Clinical Services
Published Date: June 2024































