|
Getting your Trinity Audio player ready...
|
The third and central phase is the execution phase. In this explanatory article, we highlight the roles of key teams and outline the essential documentation that guides the process – helping Sponsors and CROs understand what to expect as the work gains momentum.
Read the previous article about the preparation and setup phase in clinical trials.
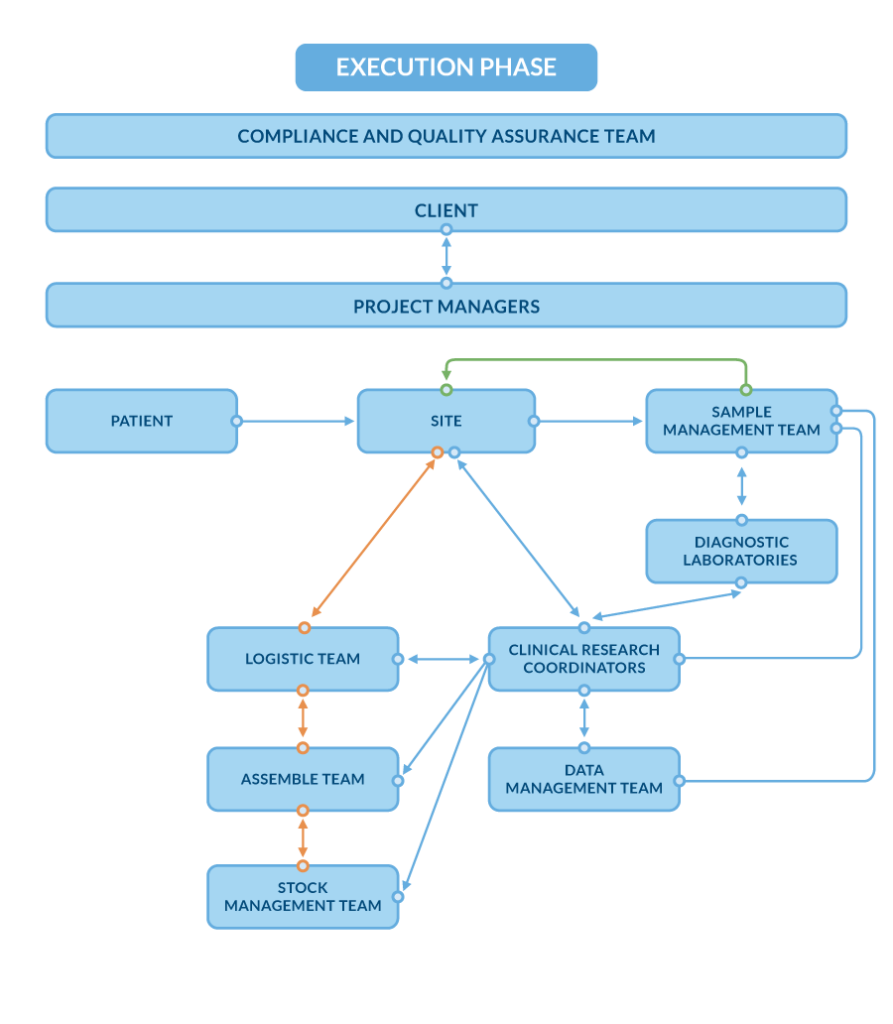
Sample Journey in the Execution Phase at Central Lab – The Most Common Scenario
There are many possible scenarios for how a sample moves through the Execution Phase, depending on the study design. Below is the most common path, showing how samples are handled from collection at the site to final data reporting.
- Sample collection at the site
Biological samples (e.g., blood, urine) are collected from trial participants in accordance with the study protocol. The sampling sets (tubes, labels, instructions, etc.) are prepared on the site request by Assemble Team and provided in advance by the Logistic Team from the Central Lab.
- Preparation and shipment from the site
After collection, samples are packed following specific shipping instructions, including temperature requirements (e.g. on dry ice). The Logistics Team oversees each step of the shipment process – from the site to the central laboratory – ensuring that no samples are lost, delayed, or compromised.
- Sample receipt and registration at the Central Lab
Upon arrival at the Central Lab, the Sample Management Team performs quality checks (e.g., data compliance, sample integrity, transport conditions). Each sample is registered and prepared for further analysis.
- Laboratory testing in the diagnostic lab
Samples are forwarded to the appropriate diagnostic/bioanalytical laboratory for testing, performed according to the study protocol requirements.
- Storage of samples & leftovers
If requested by the client or defined in the study protocol, any remaining sample material after analysis can be transferred back to the Sample Management department for storage under deep-frozen conditions. The team maintains accurate storage records and ensures availability in case retesting is required. Samples can be stored for as long as needed, in line with the protocol and client-specific requirements.
- Transfer of results
Once testing is completed, the results are reviewed and verified by the Clinical Research Coordinators to confirm compliance with the study protocol and quality standards.
- Reporting of results to sites
The Clinical Research Coordinators deliver results to the sites – most often through secure web portals or encrypted email. They also manage ongoing communication with site staff to ensure clarity and timely updates.

Teams responsibilities
The smooth operation of the Execution Phase in Central Laboratory depends on the collaboration of multiple specialized teams. Each group plays a distinct role in ensuring that clinical trial activities – from sample management to data delivery – are performed efficiently, accurately, and in full compliance with study protocols.
Project Management Team
Project Managers oversee all aspects of ongoing project phases, acting as the primary link between the Central Lab, clinical sites, and the sponsor or CRO. Often serving as the single point of contact, they coordinate timelines, monitor project milestones, and ensure that all deliverables remain on track.
Read more about the role of Project Manager.
Clinical Research Coordinators
Clinical Research Coordinators oversee the flow of laboratory data, ensuring that results are delivered promptly to investigational sites through secure web portals or encrypted email. They serve as a key communication link between the laboratory and the sites and work closely with the Data Management Team throughout the study to resolve data discrepancies as part of the daily data reconciliation process. They also ensure that all results align with protocol requirements and meet regulatory standards.
Assemble and Stock Management Team
This team is responsible for preparing and dispatching all sampling materials to clinical sites – typically within 10 working days. They manage inventory levels, provide support related to collections sets (kits) documentation, and ensure the consistent availability of tubes and auxiliary supplies throughout the study.
Logistic Team
They supervise the shipment processes to sites and performing laboratories making sure that samples and collection sets are delivered on time, under proper conditions, and without damage. The team closely monitors shipping timelines and temperature requirements at every stage, ensuring that all materials arrive under the conditions specified in the protocol.

Sample Management Team:
This team manages both the pre-analytical and post-analytical phases of each visit. They receive and inspect incoming samples, register them, checked quality and prepare them for testing. After analysis, if requested by the client or defined in the study protocol, samples can be returned for long-term storage at our deep-frozen facilities, where they are kept under monitored conditions for the duration of the study. If not, the team designs a backup aliquot, and the remaining material is usually disposed of.
Read more about the role of Sample Management Department.
Data Management Team:
The Data Management Team plays a vital role in ensuring the success and reliability of clinical trials. They are responsible for the accurate collection, storage, and analysis of study data – safeguarding its integrity, quality, and consistency throughout the trial.
Compliance & Quality Assurance Team:
This team ensures that every process across the Central Lab meets the highest regulatory compliance and internal quality standards. Through regular audits, documentation reviews, and process evaluations, they maintain alignment with Good Clinical Practice (GCP), Good Clinical Laboratory Practice (GCLP) guidelines and uphold quality assurance across all project stages.
Tools and services used in execution phase at Central Lab
Efficient execution of clinical trials relies not only on well-coordinated teams but also on specialized tools and support services.
CTMLS – Clinical Trial Management Laboratory System
Recognizing the increasing demands and specific requirements in the field of clinical trials, our Data Management Team has developed a valuable tool for both internal use and site utilization: an in-house developed database for clinical trial projects. The LabOne software offers a range of functionalities, including project management, evidence collection, trial participant and data management, and real-time connectivity with laboratory information systems.
Additionally, electronic data transfer is conducted according to the specifications set during the contracting phase and outlined in the electronic data transfer agreement, ensuring seamless integration and compliance with established standards.
In clinical trials, data collected from central labs often represents a significant portion of the overall dataset, with lab results frequently contributing up to 60-70% of the data used in regulatory submissions and analysis. This underscores the importance of rigorous data management and adherence to quality standards to ensure that trial outcomes are based on reliable and precise information.
Dry Ice shipment
Medicover MICS also provides its own dry ice solutions, tailored specifically for biosamples that require frozen transport. On request, dry ice replenishment can be arranged directly for sites, helping them maintain proper storage and shipment conditions. Read more about shipping lab samples in dry ice.
Looking for a Central Lab Partner?
to explore how we can support your next clinical trial.
FAQ – Execution phase at Central Lab
1. What happens during the clinical trials execution phase at Central Lab?
The execution phase begins once sample collection starts at clinical sites. It includes logistical coordination, sample shipment, lab testing, data verification, and results reporting. Key activities include receiving and registering samples, ensuring quality control, conducting diagnostic testing, and securely delivering validated results to sites.
2. Which teams are involved in the execution phase and what are their roles?
Several specialized teams ensure smooth execution:
– Project Managers coordinate the entire process and serve as the main point of contact.
– Logistics ensures timely and temperature-controlled sample shipments.
– Sample Management handles quality checks, registration, and long-term frozen storage.
– Clinical Research Coordinators manage data flow and site communication.
– Data Management ensures accuracy and timely transfer of lab data.
– Quality Assurance monitors compliance with regulatory and internal standards.
3. How is sample integrity maintained during transport and processing?
Samples are shipped following strict protocols, often on dry ice, and monitored for temperature compliance. Medicover MICS offers dry ice replenishment to maintain stability during transport. On arrival, samples undergo quality checks for labeling, volume, condition, and documentation before being prepared for testing.
4. How are laboratory results processed and reported to the sites?
After testing, results are reviewed for completeness by the Sample Management Team and verified by Clinical Research Coordinators. Final data is delivered to the sites via secure web portals or encrypted email. The process ensures timely access to results while maintaining compliance with the study protocol.
5. What systems and tools support the execution phase at Central Lab?
Medicover MICS uses LabOne, a CTMLS platform developed in-house to support trial management, trial participant tracking, and data flow. It enables real-time connectivity with lab systems and ensures accurate, compliant data transfer. This technology enhances visibility, reduces errors, and helps sponsors monitor trial progress.
































