|
Getting your Trinity Audio player ready...
|
Efficient collaboration with a central laboratory begins with a strong foundation. This first article in our series explores the critical preparation and setup phase that influences study timelines, logistics, and data integrity. We highlight the roles of key teams and outline the essential documentation that guides this process, helping Sponsors and CROs understand what to expect from the very beginning.
Before initiation of cooperation with Central Lab
Before collaborating with a central laboratory, the groundwork is laid by key trial documents and decisions made at the Sponsor or CRO level. The Study Protocol plays a central role, defining the laboratory scope and guiding all subsequent operational planning.
A clinical study protocol is a comprehensive document that defines the framework of a clinical trial. It includes the trial’s objectives, detailed design, and methodology, as well as statistical considerations and organizational aspects. The protocol sets out the inclusion and exclusion criteria for participants, the specific assessments to be performed, the data collection and management procedures, and safety and ethical considerations. This foundational document ensures all parties have a clear and consistent understanding of the study’s purpose, scope, and operational requirements.
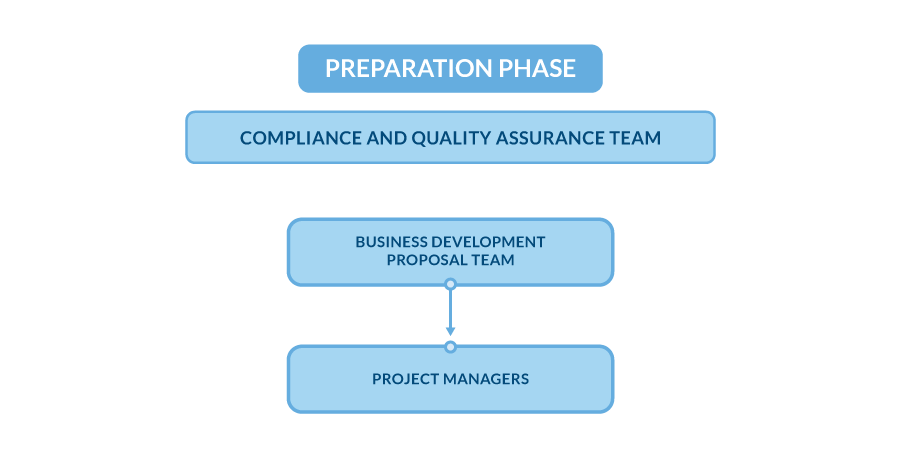
Preparation phase
This is the first phase of collaboration with the Central Lab. During this phase, the Central Lab reviews the provided Study Protocol to fully understand the laboratory requirements for the trial. This review encompasses the identification of required tests and their specific reporting parameters. Additionally, this phase involves revisiting and clarifying key study assumptions, such as clinical site locations, projected timelines, and regulatory requirements. These factors are essential for accurate planning and ensure that the laboratory’s operational activities are aligned with the study’s needs.
Seamless Transition from Commercial to Operational Teams at the Central Lab:
- Analytical Project Manager Assignment
A dedicated Analytical Project Manager is assigned to oversee the new trial. This person becomes the main point of contact and coordinates the project’s initial stages.
- Handover from Business Development to Operational Teams
Agreements and discussions with the Sponsor or CRO are formally transitioned to the operational team, led by Project Managers. With the budget approved and contracts signed, the Central Lab is ready to begin executing the project.
- Engagement of the Compliance and Quality Assurance (CQA) Team
The CQA team is actively involved from the start, maintaining continuous oversight and ensuring consistent quality and compliance across all departments. They serve as both supervisors and advisors throughout the project.
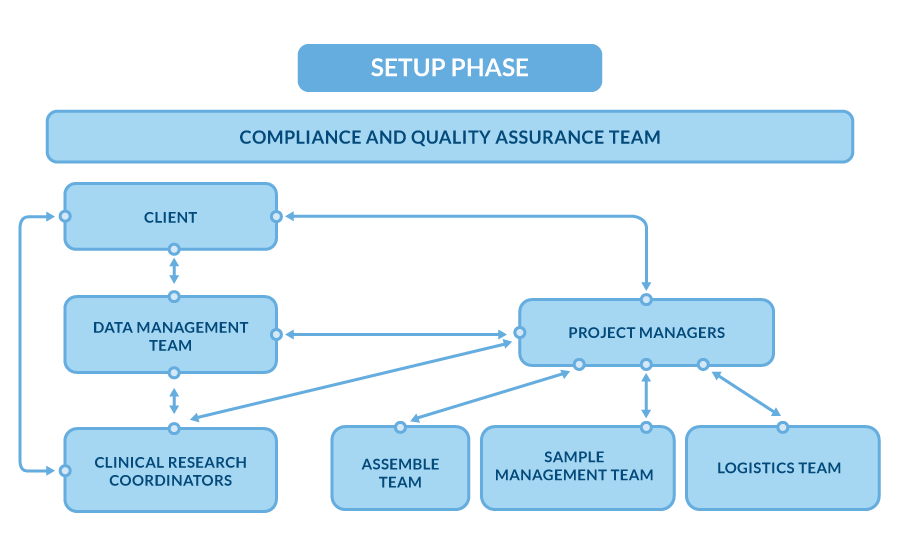
Study Setup phase
Once the preparation phase is complete, with the study team assigned and the project formally handed over, the study setup phase begins. In this phase, the Central Lab team translates the protocol requirements into detailed, actionable documentation and workflows. These materials guide the day-to-day execution of the project, ensuring all operational aspects are aligned with the study’s needs.
Key Study Documents
The following documents are prepared in strict adherence to the Study Protocol and regulatory requirements:
- Responsibility Log
This log outlines the roles and responsibilities of all parties involved in the trial. It ensures accountability and transparency by tracking delegated responsibilities, supporting regulatory compliance, and addressing any issues that arise during the study.
- Analytical Plan – A structured outline that defines laboratory activities based on the Study Protocol and the signed budget agreement. It includes:
- Roles of laboratory staff
- Study visit structure and number
- Inclusion/exclusion criteria and population definitions
- Biospecimen types (e.g., blood, plasma, tissue)
- Collection, labeling, shipping, and storage requirements
- Assay details, including required tests, methods, and timelines
- Sample collection set design
- Format and timing of lab result reports
The Analytical Plan ensures that all laboratory processes are aligned with the Study Protocol and must be approved by the Sponsor or CRO.
- Laboratory Requisition Form (LRF)
A document used to request laboratory testing and analysis of samples. It captures essential information such as participant details, the specific tests required, and study parameters. The LRF ensures samples are processed correctly and according to the protocol, supporting data accuracy and consistency. It also serves as a communication tool between the clinical site and the laboratory.
- Laboratory Manual
A vital resource for the site investigational team, the Laboratory Manual guides laboratory-related activities throughout the study. It details procedures for sample collection, processing, storage, shipment, and turnaround time (TAT) reporting. Aligned with Good Clinical Practice (GCP) standards and the Study Protocol, the manual ensures consistency and quality across all laboratory processes.
Once all the study documents are finalized and approved by the Sponsor or CRO, the following setup steps are undertaken:
Database Setup
The laboratory database, dedicated to managing clinical trial data, is configured based on the Study Protocol and contractual agreements. This ensures that the system is ready to handle and securely store project data throughout the study.
Preparation of Collection Sets
The Assembly Team prepares the collection sets in accordance with the specific requirements of the study, ensuring that all materials and components are tailored to the trial’s needs.
The design of each collection set is guided by the Analytical Plan to ensure all study-specific testing requirements are met. This process involves selecting appropriate tubes and materials based on the type of biological material to be collected, packaging considerations, and transportation conditions, including temperature requirements. By addressing these factors during the design phase, the collection sets are optimized to minimize pre-analytical errors and maintain sample integrity throughout transport.
Key features of the collection setup include:
- customization to align with the Study Protocol and visit schedule,
- pre-formatted air waybills (AWBs) to streamline documentation,
- tailored laboratory requisition forms,
- pre-labeled collection tubes to reduce labeling errors,
- a detailed Laboratory Manual with comprehensive specimen collection and handling instructions.
Quality Control of Collection Sets
The Logistics Team performs an initial quality control check on 10% of the collection sets to ensure they meet all study requirements and regulatory standards. If any discrepancies are found, a full (100%) quality control check is conducted to guarantee accuracy and completeness before shipment to clinical sites.
Shipment by the Logistics Team
The Logistics Team manages the secure transportation and storage of collection sets, ensuring timely and controlled delivery to clinical trial sites worldwide. This process guarantees that the sets arrive before the site initiation visit, so all materials are available for the study to start on schedule.
Summary
The setup phase translates protocol requirements into practical, actionable processes that are crucial for a smooth study launch. With all key documents finalized and shipments of collection sets arranged, the project is now poised to move into the Execution Phase. In our next article, we will explore this phase in detail, highlighting how the Central Lab manages daily operations, sample handling, and data delivery throughout the study.
FAQs – Preparation and Setup Phase with a Central Lab
1. What are key activities in the setup phase at central lab?
The setup phase focuses on translating the Study Protocol into practical processes and documentation. Key activities include preparing the Responsibility Log, Analytical Plan, Laboratory Manual, and Laboratory Requisition Forms. The central lab also designs and assembles study-specific collection sets and sets up the database to manage clinical trial data securely.
2. How long does the setup phase take at a central lab?
The duration of the setup phase can vary depending on the study’s complexity and the sponsor’s requirements but typically takes between 4 and 8 weeks. This phase includes document preparation, quality control of collection sets, database setup, and shipment planning to ensure all materials arrive at sites on time.
3. What quality control measures are taken during the setup phase?
Initial quality control (QC) is performed on 10% of the prepared collection sets to ensure accuracy, completeness, and compliance with study requirements. If any discrepancies are found, a full 100% QC check is carried out to guarantee that the collection sets meet the required standards before shipping.
4. How are study-specific requirements incorporated into the central lab setup?
Study-specific requirements are captured in the Analytical Plan and reflected in the customization of collection sets, labeling, and laboratory documentation. These include visit schedules, biospecimen types, collection and shipping conditions, and testing parameters, ensuring that the central lab processes align precisely with the protocol and regulatory standards.
5. What challenges can arise during the setup phase with a central lab?
Common challenges include incomplete or evolving protocol details, unclear study assumptions, and aligning laboratory processes with sponsor timelines. Effective communication and thorough review of all study documentation help address these issues and ensure operational readiness.
6. How can sponsor and CROs ensures a smooth transition to the execution phase?
Sponsors and CROs can support a smooth transition by providing timely and accurate study documents, engaging in clear communication with the central lab, and approving the final documentation promptly. This collaborative approach ensures that operational teams can launch the study with confidence.































