|
Getting your Trinity Audio player ready...
|
AI is moving from experimentation to regulation
Artificial intelligence is already present in clinical trials. What is changing now is how AI regulation in clinical trials is evolving and how its use is being structured. Currently, AI supports data analysis, feasibility assessments, documentation and operational decision-making. The key shift is not the presence of AI, but how it is being treated.
Regulators are beginning to move from observing and discussing AI to actively structure how it should be used. The recent plan published by the European medicines regulatory network shows that AI is entering a regulated environment. AI is no longer just a tool supporting clinical trials. It is becoming part of the regulated system.
What EMA actually announced?
The Network Data Steering Group (NDSG), operating within the European medicines regulatory network (EMRN), published a multi-year workplan for 2026–2028 focused on data and artificial intelligence.
Importantly, this is not a single guideline. It is a structured framework describing how AI will be gradually integrated into medicines regulation. The plan is built around three main areas:
- Guidance, policy and product support
Focus on developing AI-related guidance across the medicines lifecycle, ensuring alignment with the AI Act and GDPR, and supporting regulatory decision-making processes. - Tools and innovation
Development and deployment of AI tools within the regulatory network, alongside the creation of a framework for their consistent and controlled use. - Collaboration and change management
Building AI literacy, training stakeholders, and creating a coordinated approach across European agencies and partners.
Together, these elements show that regulators are not only defining rules, but also building the infrastructure required to use AI in a controlled way.
The timeline of AI regulation in clinical trials
The workplan outlines a clear sequence of milestones rather than a single regulatory release.
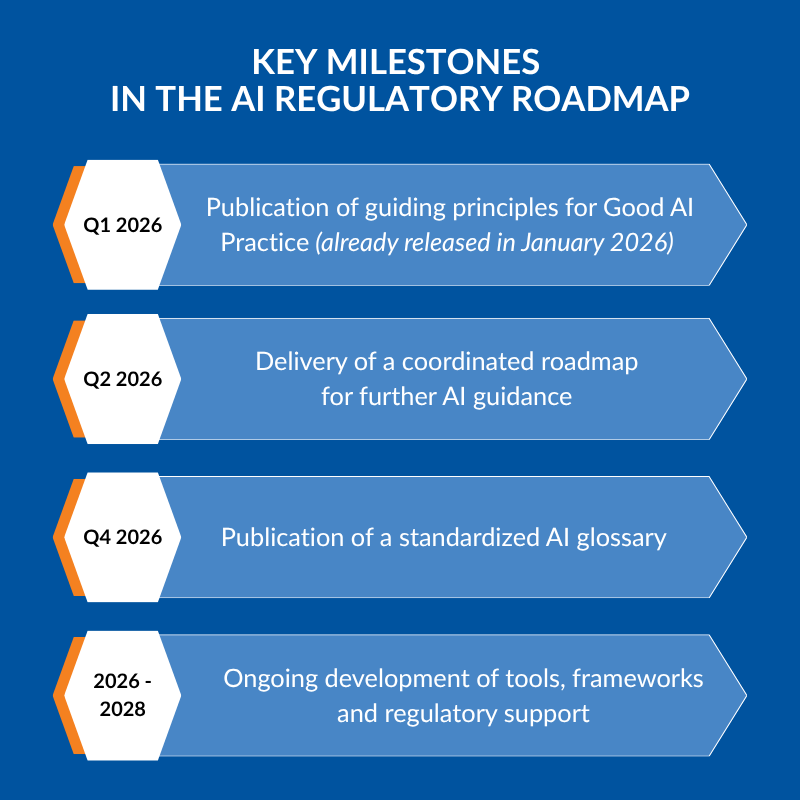
Publication of guiding principles for Good AI Practice. This confirms that the regulatory shift is already underway, not just planned.
The key shift: AI must be auditable
The most important signal from the plan is not the timeline itself, but the direction. AI is expected to operate within a controlled and compliant environment. This includes clear focus on risk management, data protection, ethical considerations, and emerging expectations around AI governance in clinical trials.
This direction is already reflected in the Guiding Principles for Good AI Practice in Drug Development, published in January 2026, which emphasize transparency, governance and lifecycle oversight of AI systems. In practice, this means that AI must be:
- controlled
- repeatable
- traceable
- documented
This is a significant shift. AI is no longer treated as an optional support tool, but as part of regulated processes that may need to be justified, reviewed and audited. AI is being treated like any other critical process in clinical trials.
What this means in practice for sponsors and CROs
For organisations involved in clinical trials, this shift introduces new expectations around AI compliance in clinical research and has direct operational implications. In practice, this means that organisations need to:
- define how AI is used within processes (e.g. SOPs) to ensure consistent and compliant use
- validate AI tools before they are implemented
- establish clear data governance frameworks
- document decisions supported by AI
- prepare for audit scenarios involving AI use
These expectations will increasingly apply across study setup, data handling and operational decision-making. AI regulation in clinical trials will not replace other regulatory requirements. It will become part of them.
Conclusion
AI regulation in clinical trials is not a future concept. It is already part of daily operations. What is changing is the level of control and accountability expected around its use. Regulators are moving quickly to define how AI should be applied, and these expectations will continue to evolve over the coming years. The question is no longer whether to use AI in clinical trials, but how to use it in a controlled and compliant way.
Looking to apply AI in a regulated environment? Request a quote to explore how we can support your study.
FAQ about AI regulation in clinical trials
1. Is AI regulation in clinical trials already in place?
AI regulation in clinical trials is still evolving, but regulators are actively building frameworks that define how AI should be used, assessed and controlled.
2. What is the purpose of the EMA AI workplan?
The EMA AI workplan aims to establish a structured approach to AI in medicines regulation, including guidance development, governance models and implementation of AI tools.
3. What does “auditable AI” mean in clinical trials?
Auditable AI means that AI-driven outputs and decisions must be explainable, documented and defensible. This is a key requirement within emerging AI compliance in clinical research.
4. How does AI governance apply to clinical trials?
AI governance in clinical trials refers to defining how AI is used, controlled and monitored within processes. It includes policies, validation approaches and oversight of AI-supported decisions.
5. Will AI tools require validation in clinical research?
Yes, expectations are growing that AI tools used in clinical trials will need to be validated, especially when they impact data quality, analysis or decision-making processes.
6. What are the main AI audit requirements in clinical trials?
AI audit requirements in clinical trials will likely focus on documentation, traceability, validation and transparency of AI use, ensuring compliance with regulatory expectations.
References
- Network Data Steering Group Workplan 2026–2028 for Data and Artificial Intelligence in Medicines Regulation, European Medicines Agency (EMA), accessed March 2026.
- Guiding Principles of Good AI Practice in Drug Development, European Medicines Agency (EMA), accessed March 2026.
- Artificial Intelligence Act (EU AI Act), European Commission, accessed March 2026.