About
Medicover Integrated Clinical Services
We offer comprehensive clinical research one-stop-shop solution for Pharma, Biotech and CRO companies.




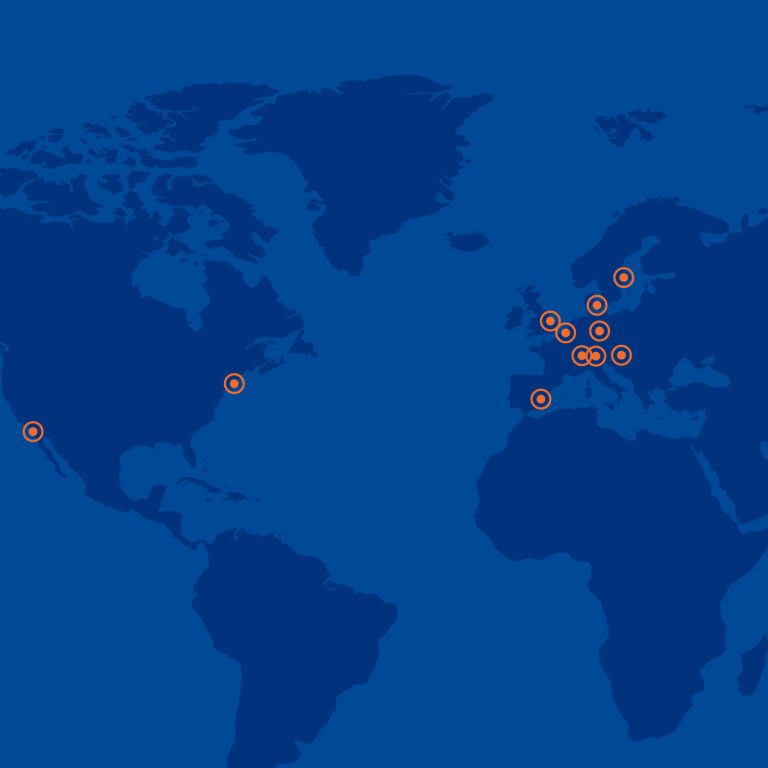
We provide globally Integrated Clinical Development Solutions
Medicover Integrated Clinical Services is a specialized business unit of Medicover AB, a Swedish multinational healthcare company. MICS uses the vast network of Medicover hospitals, clinics and labs in Europe and India, and its global network of partners to support the development of innovative drugs and therapies, and bring them to the market.
Our activities support the entire spectrum of drug and therapy development activities starting from Biomarker Discovery to Market Launch. Contact us to learn more about how we can support your research, development and commercialization efforts to bring your innovative product to market.
Global Integrated Clinical Development Solutions
Medicover Integrated Clinical Services brings innovative therapies and diagnostics to the market. We are focusing on clinical applications and clients from pharmaceuticals, diagnostics biopharma, biotech, contract research organisations (CROs), biobanks, and other players.
Our services are organized under four business lines: Central Lab Services, Site Management Organization, Precision Medicine and Companion Diagnostics.
Central Lab
Services
Comprehensive solutions to coordinate every aspect of clinical trial laboratory services. We combine our professional experience and operational excellence to support clinical trial sponsors and CROs starting from study setup to the final submission of laboratory results.
Companion
Diagnostics
We excel in providing tailored solutions to enhance personalized patient care. Our expertise spans the development and implementation of cutting-edge diagnostic tools that complement specific therapies.
Precision
Medicine
Complete multi-omics services to support targeted therapy and drug development programs. We provide end-to-end services including biomarker discovery, target validation, CDx assay development, commercialization, and regulatory services.
Site Management
Organisation
With access to major clinical study sites and a large network of more than 100 clinical centers, we successfully integrate digitization and innovative solutions supporting patient recruitment and the whole clinical trial journey.
Compliance and Quality Assurance

The Compliance and Quality Assurance (CQA) Department of Medicover Integrated Clinical Services is independent from operational departments and reports directly to our Managing Director. The CQA Department is responsible to ensure that the Organisation adheres to relevant laws, regulations, and internal policies and helps safeguard the organization’s reputation and to promote a culture of integrity and ethical conduct within the Company.
The CQA personnel keep continuous contact with each MICS unit and department to ensure the same level of compliance, quality and consistency across all our units. We continually monitor our service and operations to assure that the highest level of compliance and quality is maintained.
We have implemented an Integrated Quality Management System based on the well-recognized international quality standards and regulatory guidelines such as Good Clinical Practices (ICH GCP), Good Clinical Laboratory Practices (GCLP), ISO 15189:2022, ISO 17025:2017. Additionally, our Clinical Trials Laboratory Management System (CTLMS) is compliant with Title 21 Part 11 of the Code of Federal Regulations (21CFR Part11), which applies to electronic records and electronic signatures.
We also invest significant time and resources in our employee training and operations improvement programs.
Our integrated quality management system includes:
Work with us
We are dynamically growing, expanding our worldwide capabilisties. We are committed to fostering a globally diverse workforce and a companywide culture that encourages excellence, leadership, innovation and a balance between our personal and professional lives.
Join our team and actively contribute to the advancement of innovative medical research. We will work together to challenge the status quo and make health care better for more people, in more ways than ever. You will be part of culture that prizes innovation that impacts quality, accessibility and affordability of care across the world.
Current vacancies
Study Nurse
Localisation: Munich, Germany
Study Coordinator
Localisation: Munich, Germany
Corporate news
Explore the zone for the latest updates from Medicover Integrated Clinical Services (MICS). Stay informed about our collaborations, acquisitions, and upcoming events organized by us.